DK Science: Gases
Gases are all around us, but although many, such as perfume, can be smelt, most gases are invisible. Like liquids, gases can flow but, unlike solids or liquids, gases will not stay where they are put. They have no set shape or volume, and they expand in every direction to fill completely whatever container they are put into. If the container has no lid, the gas escapes.
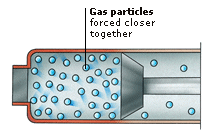
Gas particles move around at over 1,600 km (1,000 miles) per hour. The particles are widely spaced and can move freely in any direction. Gases can spread out to fill whatever container they are put into. When gas particles collide, the forces between them are not strong enough to keep them together – instead they bounce apart.

Vapour is a gas that has evaporated from a liquid before the liquid has reached its boiling point. Water, for example, boils to form a gas at 100°C (212°F). But, even at much lower temperatures, some water particles escape from the liquid to form a gas, called vapour, that mixes with the air. When vapour cools slightly, the gas forms droplets seen as mist.
In hot air balloons, a burner heats the air inside. This causes the particles of air to gain more energy and so they move faster and farther apart from one another, pushing at the sides of the balloon. Heat always causes gases to expand. If you left a balloon near a fire, the air inside could expand so much that the balloon would burst.
Why does a champagne cork explode out of a shaken bottle? The champagne inside the bottle contains lots of tiny bubbles of gas. Shaking the bottle releases the gas, and the high-speed gas particles bang against the cork. This creates an enormous pressure on the cork, and eventually forces the cork out of the bottle.