Weather: Moisture and Humidity
Moisture and Humidity
In all my years of teaching and talking about the weather, I have never encountered a concept more difficult to grasp than humidity. In fact, if you make it through, the rest will be a breeze.
Let's start off by recognizing that water can exist in three different states: solid, liquid, and vapor. All states consist of the molecule H2O; the only difference concerns the spacing of the molecules. The following figure depicts these different states. As a solid, the water molecules are closest together. As a vapor, the molecules are farthest apart. So the whole humidity story hinges on what makes these molecules drift apart, or come together. Any guesses?
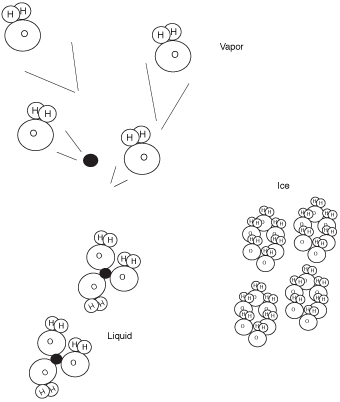
Change of phase.
How about temperature? Think of the molecules as popcorn in a popper. When the heat is first turned on, the kernels are just sitting there, solidly, on the bottom of the popper. But then, as heat is added and the temperature increases, the kernels start popping, and the popped corn moves around. Like popcorn, water vapor molecules are bouncing around, thanks to heat in the atmosphere. The water molecules are far enough apart that the vapor is invisible. The vapor itself comes from the water that is evaporated from the earth's surface, including oceans and lakes.
If the heat is reduced, the temperature lowers, and the molecules no longer move around as much. They stick. They become visible, turning into water. The water can take the form of a visible plume of steam, like a cloud, or just raindrops that form pools of water on the ground. If the temperature lowers even more, the molecules become closer. Eventually, you can even walk—or at least skate—on that water. It turns to ice. So the whole process is a function of temperature.
Weather-Speak
Relative humidity is the ratio between the actual amount of water vapor present to the capacity that the air has at a particular moment. That capacity is a function of temperature.
If the temperature is warm, the atmosphere has a greater capacity to hold water in its vaporous state than if it were cold. During the winter months, the water-vapor content of the atmosphere is as much as four times greater in the tropics than it is in mid-latitudes locations such as New York, Boston, or Chicago. In the summertime, the differences become less.
Vapor can change into visible water in one other way: Vapor molecules can also come closer together if more vapor is tossed into the mix. As more molecules congregate, they get a little cozy with each other and eventually transform into visible water. So even if the temperature stays the same, the molecules can be crowded and then change over to regular water.

Humidity factors. As the air reaches its capacity of water vapor, the excess spills out as liquid water.
Let's combine both concepts. For any given temperature, the air has a particular capacity for water vapor. If that capacity is exceeded, the excess spills out as liquid water. The concept is similar to filling a glass with soda. If the glass is full, pouring more soda into the glass (or somehow shrinking the glass) will cause the soda to spill over the sides of the glass. In the atmosphere, that spillage is made possible by the addition of more water vapor, beyond the particular capacity, or by lowering the capacity through lowering of temperature.
This may be the right moment for a real popcorn and soda break! But this is as difficult as it gets. The rest just builds on the basic concept. The figure to the right shows these concepts.
A Sense of Humid
Braving the Elements
Here's a little quiz: What was the most deadly weather disaster in New England history? The blizzard of 1888? Not exactly. How about the big floods of 1955? No, try again. The Great New England Hurricane of 1938? Wrong again. It was actually the heat wave of 1911. For nearly half the month of July, the thermometer soared into the 90s and low 100s. The humidity also increased, and the combination of heat and humidity caused at least 2,000 deaths during that month alone. A similar heat wave took the lives of more than 700 people during July 1995 in Chicago. Across the entire United States, about 1,250 deaths were attributed to the 1980 heat wave. Overall, more than 20,000 deaths due to heat waves have occurred in the United States since the mid-1930s.
Weather-Speak
Dew point is the temperature at which the vapor in the atmosphere becomes liquid. The point at which the atmosphere is totally filled is called saturation. When it is filled to overflowing, the condition is called condensation.
Relative humidity becomes the ratio between the actual amount of water vapor present to the capacity that the air has at a particular moment. Just to be an optimist, if the glass is half-filled, the relative humidity is 50 percent. If the glass is three-quarters filled, the relative humidity is 75 percent. Again, the relative humidity will depend on how tall a glass might be (capacity) and how full that glass might be. The relative humidity will increase if more vapor collects in the atmosphere, or if the temperature is lowered. If the water-vapor content remains the same, the humidity becomes dependent on the temperature. An increasing temperature will lower the relative humidity. A lowered temperature will increase the relative humidity. You see this happening all the time.
For example, you go to bed at night, and the lawn and car are dry. Skies are clear, yet, in the morning, everything is wet with dew. Where did the water come from? That's easy. It didn't rain, but the temperature cooled at night, and eventually the lowered capacity matched the amount of vapor present. Additional cooling brought the capacity within the air below the amount of vapor present. The excess spilled over. Dew formed—the dew point was reached. So dew point is the temperature at which the vapor goes over to a liquid. In this whole humidity and dew-point concept, just keep thinking of a glass getting smaller and smaller. Eventually it matches the level of its contents, and finally it spills over.
The point at which the glass is totally filled is called saturation. When it is filled to overflowing, the condition is called condensation. So when saturation is reached, the temperature equals the dew point and the relative humidity becomes 100 percent. Any increase in water vapor, or decrease in temperature, will result in condensation.
Is It the Heat or the Humidity?
When it comes to what makes you most uncomfortable, it's probably a combination of both heat and humidity, but the relative humidity alone is the poorest indicator of comfort. For example, if the temperature lowers, the humidity will automatically increase and could reach 100 percent. But if the temperature is low enough, nobody seems terribly uncomfortable. Then there are days when the relative humidity checks in at a seemingly comfortable 40 percent, but the air is stifling. During those days, the temperature reaches close to or over 90 degrees in the afternoon. Amazingly, in the morning on such a day, the relative humidity would be higher, but you probably wouldn't be complaining, because the temperature would be lower in the morning. Comfort levels seem to be a combination of both temperature and humidity, with the moisture content of the air playing a huge role. The dew point is an excellent indicator of comfort.
The dew point reflects the water vapor content in the air, and that water vapor influences the natural cooling mechanism of our bodies: sweating.
When the weather heats up, a gland in our brain called the hypothalamus tries to regulate the body temperature by setting off our 10 million sweat glands. Water, in the form of perspiration, springs up on our bodies. When that moisture evaporates, we cool down because the process of evaporation always extracts heat. Why do you think your skin feels cool when alcohol is rubbed on it? Because alcohol evaporates very rapidly.
Weather-Speak
The heat index describes what the air feels like given the combination of temperature and humidity.
If the air is filled with water vapor, it can't hold much additional water. So when the dew point is high and the water vapor content is high, less perspiration can evaporate into the air. Our bodies can't cool as effectively. Dew point readings in the 60s indicate moderately uncomfortable weather. When the dew point climbs over 70 degrees, the air becomes very uncomfortable, and as it approaches 80 degrees, it becomes tough to breathe, even for normally healthy people. Of course, when the temperature becomes high, the effect of a high dew point is magnified. Th e high temperature makes our bodies perspire, and the high dew point works against the natural cooling mechanism.
In hot weather, rapid water and salt loss in your body can cause a chemical imbalance that leads to heat cramps. Additional water loss and increased temperature brings on heat exhaustion, which causes fatigue, headache, nausea, and even fainting. If the body temperature reaches 106 degrees, heat stroke becomes possible. The body completely shuts down, and this can be fatal.
A heat index is commonly used to describe comfort levels. It combines temperature and humidity. The table below shows that index and its effects. The index is represented by an apparent temperature, or what the air may feel like to most people because of the combination of temperature and moisture. That apparent temperature is derived by combining the air temperature with the relative humidity. The relationship is shown in the following figure.

Heat index.
For example, if the temperature is 90 degrees and the relative humidity is 50 percent, the heat index, or apparent temperature, is 93 degrees. At that level, the following table tells us that sunstroke, heat cramps, and exhaustion become possible with prolonged exposure and physical activity. What level of activity is dangerous really depends on the age and health of an individual, but for most people, an apparent temperature of 90 degrees sends a warning to slow down.
You may not be roaming the countryside with charts and tables in your pocket, but if you plan to be physically active, you can at least keep an eye or ear to the weather people when they give the dew point. A dew point of 70 or more will deliver a heat index of near or over 90 degrees with even modest heating. Dew points that soar through the 70s will likely give a heat index of 100 or more. Dew points of 80 are just plain oppressive to everyone. Let that be an excuse to sit under a shady tree or go to an air-conditioned mall.
Braving the Elements
The hottest temperature ever recorded in the United States occurred on July 10, 1913, at Greenland Ranch, California, where the temperature reached 134 degrees. In Libya, the world's highest temperature was recorded at 136 degrees. Based on an average annual temperature of 77.8 degrees, Key West, Florida, holds the distinction of being the hottest city in the United States. Miami is the second hottest with an average of 75.9 degrees. Yet Chicago experiences more days with 90-degree temperatures than Miami. The U.S. city with the highest one-time recorded temperature is Phoenix, Arizona, with a blistering 118 degrees. In Europe, Seville takes the hot temperature honors with a 117-degree reading. And if you are planning to head for Djibouti, Djibouti (near Ethiopia), travel light. That west-African city has the distinction of being the hottest in the world, with an average temperature of 86 degrees.
Weather-Watch
Indoor environmental conditions can be just as hazardous as outdoor. During the winter, the dew point is extremely low outdoors, and when that air is brought indoors and heated to room temperature, the relative humidity becomes desert-dry. The humidity can drop to near 10 percent. The dryness strains our respiratory system and can lead to infection. Pans of water near the radiator, or humidifiers, can help. But be careful of putting too much moisture into the air. When the indoor relative humidity reaches over 60 percent, mold and mildew develop.

Excerpted from The Complete Idiot's Guide to Weather © 2002 by Mel Goldstein, Ph.D.. All rights reserved including the right of reproduction in whole or in part in any form. Used by arrangement with Alpha Books, a member of Penguin Group (USA) Inc.
