DK Science & Technology: Nuclear Energy
The energy that makes the stars shine and produces the heat inside a nuclear reactor is nuclear energy. It is produced by the strong force that holds protons and neutrons together inside atomic nuclei.
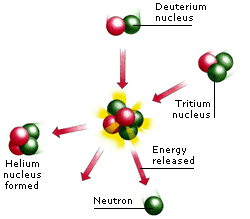
Two kinds of reaction release nuclear energy. Fusion takes place when two light nuclei combine (fuse) to make a heavier nucleus. This is the process that powers the stars. Fission takes place when an unstable nucleus of a heavy element, such as uranium, splits in two. Fission is used in nuclear power stations.
The fission of RADIOACTIVE uranium produces nuclear energy. The process is controlled by adjusting the number of neutrons produced. Control rods that absorb neutrons are inserted between the uranium fuel rods. They are raised or lowered to maintain a steady release of energy.
Strong forces act inside the nuclei of atoms. Some atoms are unstable—they decay (break down) over time, releasing energy as radiation. These atoms are radioactive. There are three main types of radiation—alpha, beta, and gamma.
Radioactive substances decay over time in a predictable way. Geologists and archaeologists can date rocks by measuring the radiation they emit. In industry, radiation is used to trace underground pipes and to kill dangerous germs on food before it is packaged. In medicine, radiation treats cancer and sterilizes medical instruments.
To a living cell, an alpha particle, beta particle, or gamma ray is like a bullet from a gun. Its energy damages molecules, disrupting the cell’s life processes. Long-term exposure to radiation can cause cancers, such as leukemia. Exposure to a single, large dose of radiation can produce radiation sickness and death. Radioactive material must be handled with great care.
Polish-born physicist Marie Curie was one of the first scientists to investigate radioactivity. She discovered the radioactive element radium. She was the first person ever to win two Nobel Prizes. Marie Curie died of leukemia caused by the radiation she worked with.